Protege Undergraduate Research Program
The protege program was started by the College of Engineering and Applied Science during the summer of 2013. The goal of the program is to make students aware of opportunities for careers in research. These opportunities are for outstanding first-year students to work with leading CEAS research faculty in their faculty labs during the summer after their first year in CEAS.
2026 Applications Closed
Protege student applications for summer 2026 are now closed. Project information below is retained for current participants.
Benefits
Participating students will receive $15/hr for 40 hrs/week for a minimum of 12 weeks and a maximum of 15 weeks in the summer. Program length will be determined by the faculty supervisor.
The opportunity allows students to determine if they wish to pursue a co-op position and career in research. At the same time, participating industrial and governmental partners have an early opportunity to recruit highly talented and motivated student researchers.
Selection Process
Each spring, first year CEAS students who performed at a high level during the autumn semester are identified by a CEAS faculty committee. Most participating students have grade point averages of 3.75. Students are interviewed to determine interest in research careers and availability to work in Cincinnati or at the location of an industrial or governmental partner over the summer.
Expectations
- Report for work at appropriate times
- Learn as much as possible about your project
- Show interest in and enthusiasm for the project
- Do good research
- Participate in final symposium
- Don't work more than forty (40) hours per week!
2026 Research Projects
Research projects that are involved with the Protege program must advance the state of knowledge or understanding in a particular field. Students do not usually define the project and are defined by the mentors. These are not make-work projects. Protege students usually contribute to an important, larger project that are usually funded by some agency or industry that are important to the company. Students and mentors must agree on the project.

Access to reliable electricity is a hidden constraint in most modern medical testing. Many molecular diagnostic instruments assume stable grid power, controlled laboratory environments, and batteries or electronics for timing and actuation. This creates barriers for deployment in settings where electricity is unreliable or unavailable, and it can limit the ability to respond quickly to infectious disease outbreaks. In parallel, the supply chain burden of batteries and certain electronic materials motivates new approaches that reduce reliance on conventional powered hardware.
This project explores an alternative design philosophy for molecular diagnostic devices based on “toy-inspired physics.” Many simple toys and novelty devices exhibit repeatable, self-sustained behaviors driven by everyday energy sources and fundamental transport processes. Examples include the drinking bird toy (Fig. A), which can produce periodic motion through coupled evaporation, condensation, and gravitational effects, and lava-lamp-like systems (Fig. B), which can generate cyclical flow patterns through buoyancy and heat transfer. These systems can behave like oscillators, producing motion with a characteristic time scale without microcontrollers, motors, or conventional electrical timing circuits.
The student will work with a research team to investigate how these physical phenomena can be harnessed to generate simple, programmable motion that can actuate mechanical elements in a diagnostic device. The work will involve observing and characterizing toy-like oscillatory behavior, relating it to underlying mechanisms such as phase change, capillarity, buoyancy-driven flow, and thermal transport, and exploring how such motion could be coupled to basic fluid handling and timing functions relevant to molecular testing. The student will gain experience in experimental measurement, design iteration, documentation in a laboratory notebook, and communicating results through short reports and a final presentation.
The long-term goal is to inform new device concepts that can perform essential diagnostic functions with minimal dependence on electricity, while remaining practical and robust. The project is intentionally exploratory and emphasizes learning how to connect fundamental physical principles to real-world constraints in global health and technology deployment.
Peripheral nerve injuries impact millions of Americans each year with many more living with the effects of traumatic nerve injuries. In severe peripheral nerve injuries, nerve pathways, connections, and the extracellular matrix (ECM) tissue surrounding nerve that lead to the sensory or motor targets are damaged. If the injury gap is larger than approximately 2 centimeters, functional recovery is extremely limited and no adequate therapies are currently available for doctors. This deeply impacts the quality of life due to the loss of connection between the central nervous system and the body’s extremities. Neurons do not have the ability or needed signals from the damaged ECM surrounding the injury site to allow guided growth of axons across the injury space. Therefore, this project will address the urgent need to engineer new biomaterials that give the proper electrical, physical, and chemical signaling to cells and neurons for guided nerve regeneration and provide functional recoveries in traumatic nerve injuries. This will be accomplished by engineering biomaterials that cells will attach to (Figure 1A,B) and possess the proper physical, electrical, and chemical signals in order to direct cell alignment and growth. We will develop these biomaterials to deliver defined, tunable signals to the cells to elicit the desired cellular outcomes (Figure 1C,D). It is hypothesized that the biomaterial, in combination with relevant cells and ECM, will functionally bridge the injury gap to restore function to injured tissues.
Uncertainty is intrinsic to many real-world decision-making problems, particularly in healthcare operations management. In this domain, decision-makers face numerous sources of uncertainty (e.g., demand, surgery duration, and length of stay). Moreover, they often have limited data or only partial information about the underlying probability distributions of uncertain factors. As a result, these distributions are difficult to characterize and are subject to ambiguity. Decision-makers must therefore navigate a trade-off between adopting an optimistic or a pessimistic attitude toward distributional ambiguity. In the optimistic case, the decision-maker focuses on favorable distributions or on fixed distributional beliefs, which may lead to optimistically biased decisions. In the pessimistic case, the focus is on hedging against least favorable distributions or worst-case scenarios, often resulting in overly conservative decisions.
The presence of multiple conflicting objectives further complicates decision-making in healthcare operations. Limited and costly resources introduce trade-offs among stakeholders with competing objectives. For example, reducing wait times for critical patients or avoiding physician overwork. Depending on the objectives a decision-maker prioritizes, these trade-offs can yield favorable outcomes (e.g., timely treatment or balanced workloads) or adverse outcomes (e.g., deterioration in patient condition or excessive physician overtime). Understanding how a decision-maker’s optimistic or pessimistic attitude affects each stakeholder is therefore essential, since scenarios that benefit one objective may simultaneously harm another due to the interplay of uncertainty and multiple criteria.
Identifying the entire spectrum of decisions across multiple, potentially conflicting objectives is often computationally expensive. When a decision-maker’s preferences across objectives are known, they can be used to reduce a multi-objective problem to a single-objective one. In practice, however, eliciting accurate and stable decision-maker preferences is notoriously difficult.
The challenges described above have been further amplified by recent advances in large language models (LLMs). Emerging multi-agent frameworks enable teams of LLM agents to collaborate, specialize, and simulate human-like social dynamics to address complex tasks. Such multi-agent LLM systems have already demonstrated significant potential in supporting complex clinical decision-making, as well as decision-making more broadly, often through structured debate among multiple agents.
Building on this paradigm, this project investigates the use of multi-agent LLM systems in healthcare operations management to support decision-making under uncertainty and multiple conflicting objectives. The goal of the project is to develop an educational game that integrates optimization models for classical healthcare operations problems, including resource allocation, staff scheduling, and appointment scheduling. In the game, participants will assume the role of decision-makers tasked with designing and evaluating strategies for resource allocation, staff scheduling, and patient flow management under uncertainty. By engaging with the game, participants will gain hands-on experience with the fundamentals of optimization and simulation, positioning the game as a powerful platform for experiential learning.
This project will be completed in the Operations Research and Data Science Lab in the Department of Mechanical and Materials Engineering. You will be part of a team working at the intersection of optimization, data science, and artificial intelligence.
Preferred skills include familiarity with mathematical optimization and algorithms, programming experience in Python, and curiosity about machine learning and AI. These skills are not required; if not already possessed, they will be developed as part of the project. Training will include an introduction to mathematical optimization, instruction in Python optimization libraries and good coding practices, and guidance in scientific writing.
Imagine a water sensor that monitors a remote river for years on a tiny battery, or a wearable that detects arrhythmia instantly without the privacy risk of streaming raw data to the cloud. This project explores analog edge intelligence, where signals are processed in the analog domain to reduce power-hungry digital compute.
You will investigate how this "Green AI" approach can be deployed in smart agriculture, ecological sensing and medical IoT to create ultra-low-power, "deploy-and-forget" systems. Your focus will be on security-aware detection: building circuits that don’t just spot sensor failures (drift/spikes), but also detect malicious spoofing, replay, or signal interference/jamming.
A goal for this project is for the student to achieve co-authorship on at least one research publication resulting from the work with the Ph.D. student.
Hardware Implementation: Circuit Design
For the hands-on component, you will build a programmable analog anomaly detector using standard electronic hardware like Op-Amps and Comparators.
- Tunable Detection: You will design a "Window Detector" that stays on the lookout for anomalies (signal value), made adjustable via RC networks or digital potentiometers.
- Spectral Analysis: You will explore analog filters to extract "signatures" (like specific tones or energy bands) that reveal whether a signal is a natural event or a security attack, such as jamming.
Deliverables:
The "Analog vs. Digital" Report: A survey on how new-age analog computing can replace "always-on" digital processors in ecological and medical sensing.
The Working Demo: A breadboarded circuit that successfully classifies signals into three categories: Normal, Contamination Anomaly, or Security Attack.
Set up and document a reproducible integrated-circuit design workflow using the SkyWater SKY130nm PDK to prepare for future tapeout research.
Side-channel analysis can infer the secret key on a device (e.g., a microcontroller, a secure chip on a credit card, or an IoT device) by analyzing power consumption when the device runs encryption algorithms, such as Advanced Standard Encryption (AES). It is one of the primary threats to the security of embedded systems.
This project aims to investigate pre-silicon side-channel analysis over hardware design, e.g., RISC-V CPU, written in Hardware Description Languages (e.g., Verilog) to identify security vulnerability of software/hardware implementation of encryption algorithms. The students in this project will have the opportunities to (1) Study research papers related to side-channel analysis (including deep learning side-channel analysis); (2) Examine different hardware designs of encryption algorithms, such as AES, written in Verilog or VHDL; (3) Learn cybersecurity knowledge and skills related to this project; (4) Have access to GPU machines in Dr. Wang’s lab for training neural networks; (5) Have access to data collection platform and hardware in Dr. Wang’s lab to collect power traces of AES encryption for pre-silicon side-channel analysis. Undergraduate researchers who previously worked with Dr. Wang have received UC undergrad research fellowship awards and have published multiple research papers.

Microplastics represesent a recalcitrant biohazrad uqiuoutous in the environment because of slow and ineffecient enzymatic degradation. Recently, PET plastic degrading enyzmes having become available following the discovery of a soil bacteria capable of using TPA as its sole carbon source, essentially living off plastic water bottle waste. While many researchers have applied this to larger plastic waste sources, microplastics represent a valuable target, because of increased surface area (relative to volume) and the difficulty removing these plastics from environmental sources like drinking and wastewater. We seek to develop an efficient enzymatic removal process that uses engineering plastic degrading “termites” consisting of rotifers engineered with bacteria in their guts capable of digesting plastic, similar to how termites digest wood.
To achieve this the Protégé participant will first assist with forming PET microparticles using a microfluidic cell we have produced for this purpose. Next, the student will feed these particles and record their digestion. The student will have the opportunity to try different plastic degrading enzymes and different bacteria within the rotifers to optimize the system. The student working on this project will work closely with Dr. Wendell and senior undergraduate students already supporting the investigation.
Voice disorders affect millions of Americans each year, often resulting from scarring of the vocal folds after injury or prolonged intubation. Current surgical options can improve symptoms but do not restore the delicate tissue mechanics needed for natural voice production. This project explores a novel extracellular matrix–derived biologic (VFLPx) developed by our lab to promote vocal fold healing and reduce fibrosis through minimally invasive delivery systems.
Summer Research Experience
Students will gain hands-on experience in:
- Tissue engineering and biomaterials characterization, including formulation of aerosolized and injectable VFLPx preparations
- Cell and molecular assays to measure fibrotic and inflammatory responses
- Preclinical model design and analysis of biomechanical and imaging data related to vocal fold tissue recovery
Why Join This Project
This interdisciplinary project bridges biomedical engineering, otolaryngology, and regenerative medicine, providing an ideal environment for undergraduates interested in translational research. Students will learn how engineered biologics move from bench to bedside—contributing to innovations that may one day make non-invasive voice restoration a clinical reality.
Vascular calcification is a regulated deposition of hydroxyapatite mineral in the arteries narrowing the arterial space. A process once considered irreversible is now believed to potentially be preventable. Vascular calcification typically occurs in the medial or intimal layers of the blood vessels. Medial calcification is the most immense type of vascular calcification mainly found in patients with kidney disease, type 2 diabetes, and has a higher cardiovascular mortality rate. Medial calcification can cause a degradation of elastin fibers.
To date, no relevant research has been done creating a 3D model of vascular calcification that can be used to assess calcification, the degradation of elastin fiber, and how calcified arteries respond and retain drug therapies. My research group has developed a 3D arterial model of vascular calcification made of porcine renal arteries. The porcine artery underwent the process of decellularization using a combination method of ion-ionic detergents, ionic detergents, and nucleases. DNA analysis and histology staining have shown removal of the porcine DNA while maintaining an intact extracellular matrix. We want to provide a proof-of-concept model of vascular calcification to standardize and improve vascular calcification research. For this protégé project, we will recellularize the porcine artery with human vascular smooth muscle cells and induce calcification as well as use a bioreactor to create a realistic disease state using mechanical artery properties.
Experimental Plan:
- Decellularize porcine arteries
- Confirm DNA removal
- Seed human vascular smooth muscle cells onto porcine scaffold.
- Induce calcification of the 3D model.
- Characterize the calcification.
Training Provided
- Aseptic technique
- Cell culture
- Potential for research publications and presentations at local and national conferences
Electric Ducted fan Research
Electrically driven fans and compressors are finding applications in everything from aircraft propulsion down to hand dryers, vacuums, and power tools. This research project will be focused on studying the noise generated from an electrically driven fan with opportunities to assist simulation and experimental efforts.
Research tasks may include
- Particle Image Velocimetry (PIV) of electric ducted fans
- Use PIV to measure velocity, turbulence, and vorticity of electric ducted fans in a wind tunnel with and without structural interaction
- Computational Fluid Dynamics and Aeroacoustics simulations of a ducted fan
- Meshing and simulating fluid flow in a ducted fan configuration in Volcano ScaLES
- Investigating the capability of aeroacoustic modules of Volcano ScaLES
Instrumentation and Data Acquisition for the Hypersonic Propulsion Wind Tunnel (HPWT)
Hypersonic flight involves flying at Mach numbers greater than Mach 5. The propulsion systems to propel aircraft at these speeds have multiple shock wave systems for compressing the incoming air for achieving combustion. We have designed and fabricated a new high-speed wind tunnel facility called the Hypersonic Propulsion Wind Tunnel (HPWT). The facility will have full optical access in the test section to enable advanced optical diagnostics internal to the propulsion flowpath with the ability to control backpressure which simulates changing conditions during hypersonic flight. The facility will include instrumentation such as pressure, temperature, and flow measurement that needs to be integrated with the wind tunnel. Additionally, a data acquisition and control code needs to be written for operation and control of the facility and recording data during testing.
This project will involve assisting with:
- Installation of the wind tunnel instrumentation and control hardware
- Development of software for wind tunnel control and data acquisition
- Assisting with experimental measurements on the HPWT and a supersonic cavity flow experiment
Aeroacoustics of Human Ears
Aeroacoustics is the study of noise generated from aerodynamic phenomena. A common source of noise, aerodynamic turbulence, is found everywhere from nature to aircraft systems. This project will involve measuring the turbulent flow over a human head and human ear across a range of conditions in a low-speed wind tunnel. This project will analyze detailed measurements of the turbulent flow, realistic acoustics in the human ear, and study flow control to reduce turbulence noise on the human ear to improve audibility in windy environments.
Initial acoustic and flow measurements have been conducted on the ear installed in a 3D-printed model head. The student involved will
- Analyze the velocity field data already acquired
- Develop designs for controlling the flow in the vicinity of the ear to reduce noise.
- Acquire additional acoustic and flow measurements with the flow control devices applied will be conducted to characterize the effectiveness and change in the acoustic source field.
Dr. Hoilett is an Assistant Professor of Biomedical Engineering. He graduated from Purdue University in 2021 with a PhD in Biomedical Engineering and from Vanderbilt University in 2014 with a Bachelor of Engineering also in Biomedical Engineering. His academic interests involve developing wearable sensors for mobile health platforms and engaging engineering design activities for undergraduate and graduate students. He has several years of experience with analog circuit design and embedded systems. His previous work includes developing a wearable opioid overdose monitor for substance use disorder patients and developing an impedance analyzer for a microfluidic blood-brain barrier model among other projects. On the education side, he has developed several lab activities for first year and junior level Biomedical Engineering students. He was instrumental in developing the Milestones Program at Purdue, a certificate-granting series of workshops providing students professional engineering skills utilized in rapid-prototyping and industrial applications. At the University of Cincinnati, Dr. Hoilett’s lab will continue developing low-cost diagnostics and sensors for improving patient-care both in high-resource and low-resource settings, tackling healthcare disparities and unmet clinical needs worldwide.

Babies born premature (approximately 10% of the population), can experience a range of neurological, ocular, respiratory, gastrointestinal and musculoskeletal impairments. For these infants, proper nutrition in the early stages of life is crucial to achieving short-term clinical milestones and long-term growth and development. The best nutrition source for premature newborns to achieve these goals is breastmilk but preserving the nutrient benefits of this complex liquid biologic tissue can be very challenging.
It has been established that there is substantial adsorption of macronutrients to the surfaces of containers, feeding tube lines, and other interfaces, a process that often reduces the total nutrient delivery to the infant. For donor milk, the nutritional loss is especially severe because there are a considerable number of steps between expression of the breast milk and its delivery to the infant. These include, pasteurizing, screening, pooling, freezing, transporting, thawing, preparing, and administering the milk. Each step offers the potential for macronutrient loss, and it is particularly detrimental to infants that require tube feeding. In order to improve clinical outcomes for at-risk infants, it is critical to ameliorate this macronutrient loss.
While others have examined single components of the milk transfer system, we propose to apply a systems-level analysis to the entire milk transfer system and identify the points where macronutrient loss is the greatest. We will then use a patented technology to develop a polymer-based lining material that will prevent macronutrient adsorption and significantly improve nutrient delivery to infants, especially those in the NICU. This project’s specific aims are:
Aim 1: Characterize the amount of macronutrient loss through current collection, storage, and handling methods utilized in the processing of freshly expressed and frozen (mother’s and pasteurized donor human milk (PDHM)) and determine critical points of loss within the system.
Aim 2: Demonstrate the ability of pharmaceutical grade polyethylene glycol (PEG), a strongly hydrophilic molecule, and calfactant (trade name Infasurf), a naturally derived surfactant, to significantly decrease nutrient loss from polymers in the milk handling system.
Aim 3: Demonstrate the successful translation of nutrient-protecting coatings in a prototype milk-transfer system.
This project is in collaboration with Cincinnati Children’s and Professor Eric Nauman’s Human Injury Research and Regenerative Technologies (HIRRT) lab.
Replace this text component with your accordion's content.

Angelman syndrome (AS) is a neurogenetic syndrome associated with severe developmental and intellectual disability. Symptoms of AS include very limited speech, difficulty sleeping, and poor motor function. AS is caused by a mutation or deletion of the UBE3A gene on the maternal chromosome 15 and is typically diagnosed in the early childhood period using genetic testing. At present, there is no cure for AS. Treatment is focused on supportive therapies and pharmaceuticals to manage seizures and reduce behavior problems. The AS community is motivated to find a cure, and several major gene therapy trials are underway to reduce or potentially reverse symptoms.
Despite increased investment in clinical trials, functional outcomes that impact the lives of people living with AS and their families remain widely variable. A major barrier to clinical trials is the lack of available outcome measures suitable for AS. Wearable devices that monitor physiological and behavioral output are promising solutions because they provide objective metrics of physiological function without requiring verbal or motoric input from the patient. However, typical wearable devices in form factors such as smartwatches and headbands are often not tolerable by AS patients. Using input from AS caregivers from extensive focus groups (N = 8) conducted by our research team, we have designed MyA, a biometric vest that will measure several key signals relevant to AS (heart rate, respiration, vocalizations, temperature, and sleep), while meeting the unique sensory and tactile needs of the AS population. For our preliminary developments, we sewed conductive fabric and conductive silicone into separate commercially available athletic vests composed of primarily nylon, polyester, or polyamide. Our preliminary measurements of the electrocardiogram demonstrate the ability to capture key biosignals using our textile prototype. We were able to measure heart rate from the electrocardiogram within 2.4 beats per minute of a commercially available reference chest strap heart rate monitor with high correlation between the two devices (r = 0.88, p < 0.05).
For the proposed project, the student researcher will work closely with a senior member of the lab to integrate audio and video capabilities into the vest. Audio and video are important behavioral inputs that allow us to understand levels and styles of communication in people with AS. We would also like to understand how people with AS interact with others and their environment, and vice versa. The student will work with the senior member of the lab to validate each sensor on the benchtop and develop code that can be integrated seamlessly into the biosensor prototype.
This project is in collaboration with Purdue University Department of Psychological Sciences.
Replace this text component with your accordion's content.
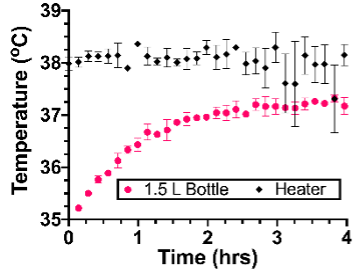
Worldwide, premature birth accounts for 30% of all neonatal deaths in children under the age of five. Neonatal hypothermia is one of the most common complications of premature birth as newborns are unable to regulate their body temperature. Hypothermia is an especially devastating complication of premature birth in low-resource settings. Over 80% of deaths due to premature birth occur in low and middle-income countries (LMICs), due to a lack of access to incubators that are critical for managing hypothermia. Furthermore, LMICs lack the necessary tools to regularly monitor the baby’s vital signs, which is critical for managing complications due to premature birth. Deaths from sudden infant death syndrome (SIDS) and accidental suffocation are silent and go unnoticed without continuous vital sign monitoring. Kangaroo mother care (KMC) is a proven intervention for combating neonatal hypothermia and involves skin-to-skin contact between the caregiver and the newborn. However, KMC places high demand on caregivers, and leaves newborns with no protection when caregivers require a break to care for themselves. Furthermore, it is very difficult to measure a baby’s vitals during KMC as the baby is swaddled tightly to the caregiver and is not accessible. As a result, KMC still leads to deaths due to SIDS or accidental suffocation as the caregiver is unaware of the baby’s vitals during KMC.
Our patented device, NeoWarm, is a disinfection wipe-friendly infant carrier that serves the need for thermal management and vital signs monitoring of premature infants during and in between KMC. NeoWarm has integrated fabric heating pads that generate heat from a lightweight, rechargeable, portable battery pack in similar fashion to an electric jacket. NeoWarm also includes sensors for heart rate, respiratory rate, blood oxygen, and temperature, to monitor the infant’s key vital signs to detect signs of hypothermia and other complications such as apnea or SIDS. Given the strength of our benchtop and preclinical studies, our present study aims to validate NeoWarm in a clinical setting with human babies. To accomplish these goals, we have the following aims:
Aim 1 (Electronic Circuit Design Aim / Option #1 for student): Develop a the second-generation NeoWarm device with integrated sensors for heart rate, respiration, blood-oxygen, and temperature. The student will help develop a professional-grade printed circuit board as well as firmware that will simultaneously track each physiological signal of interest using a microcontroller and send the resulting data to a previously developed smartphone application using Bluetooth Low-Energy.
Aim 2 (Microbiology Aim / Option #2 for student): While NeoWarm has the potential in reducing neonatal hypothermia, its role as a potential fomite is yet to be determined. Neonatal care settings pose a heightened risk of neonatal sepsis due to the vulnerable population of preterm infants with underdeveloped immune systems. Pathogens such as Enterococcus faecium, Acinetobacter baumannii and Pseudomonas aeruginosa are implicated in neonatal sepsis. Similarly, indicator organisms, such as Escherichia coli are noteworthy in neonatal sepsis due to their association with contamination and poor infection control practices in healthcare settings. To further ensure the safety of the NeoWarm prior clinical trials, a comprehensive microbial risk assessment is imperative.
The interested student can choose to work on Aim 1, Aim 2, or both.
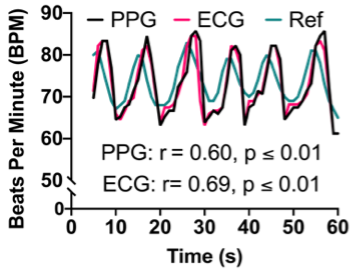

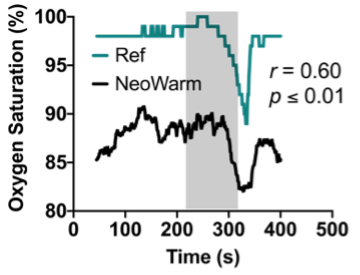
Premature infants, especially those born over 10 weeks early, are at greater risk for intraventricular hemorrhage (IVH), a condition characterized by bleeding of the ventricles due to underdeveloped blood vessels. This risk is heightened in very low birth weight (VLBW) neonates, who typically have a gestational age of less than 30 weeks and weigh less than 1.5 kg. IVH is classified into four grades. Grades III and IV are the most severe and can potentially lead to long-term developmental and motor issues, as well as an increased mortality. Transport is a known risk factor for developing IVH. However, many premature infants must be transported due to their need for specialized neonatal intensive care that may be unavailable at their birth facilities. Studies show that VLBW neonates born outside of tertiary care centers (outborn) and transported to specialized facilities have a higher incidence of severe IVH compared to those born in specialized care centers (inborn). The increased risk of developing IVH due to transport is potentially linked to the forces experienced during transport, including whole-body vibrations, translational forces, rotational moments, and excessive sound. As a result, many have endeavored to characterize these distressing forces and develop novel techniques to reduce them to acceptable levels. Some studies have suggested using a foam, air, or gel mattress inside the isolette to absorb the forces inside the isolette; however, the results were not statistically significant. As a result of this unmet clinical need, our goal is to mitigate the vibrations and noise levels experienced during all forms of transport (ground, fixed-wing, and rotary-wing), towards decreasing the incidence of IVH and improving the outcomes for premature infants worldwide.
In our preliminary work, we have documented the mechanical vibration and sound exposure experienced by a neonatal manikin during ground transport in a Type 1 ambulance provided by Cincinnati Children’s Hospital and Medical Center (CCHMC). We used 14 triaxial accelerometers, referred to as “Peapods,” and two sound level meters. The Peapods were placed on the manikin, isolette, and throughout the interior of the ambulance (Fig. 1A). The sensor on the manikin was placed on the manikin’s forehead (Fig. 1B and 1C). One sound meter was placed inside the isolette, while the other was placed on top of the isolette. The ambulance drove for 25 minutes with a mix of highway and city miles, and briefly activated the siren to simulate authentic transport scenarios. Our results indicated that the manikin experienced forces exceeding the recommended “comfortable” 0.032 g limit for the entire duration of the simulated transport event. The average noise level inside the isolette was 63.4 dBA, which is 18.4 dBA higher than the level recommended by the AAP. Given the strength of our preliminary data, our current study aims to characterize the vibrations and noises experienced during ground transport in a Type II ambulance, as well as during fixed (ambulance) and rotary wing transport (helicopter). We will then evaluate methods to mitigate the mechanical vibrations and noise levels experienced during all forms of transport. We will accomplish these goals with the following specific aims:
Aim 1: Determine forces and noise levels experienced by a neonatal manikin during a non-ambulatory transport event in a type II ambulance, during fixed-wing transport, and during rotary-wing transport. Aim 2: Determine the vibration dampening capabilities of memory foam. Aim 3: Determine the optimum soundproofing material for the transport isolette per weight of material.
Intra-abdominal hypertension (IAH) and abdominal compartment syndrome (ACS) are comorbid conditions that could result in low perfusion to the abdominal area and lower extremities, consequently negatively affecting oxygen delivery to organs, ultimately leading to organ failure, and death. Incidence rates of ACS in the literature vary, but some reports show rates as high as 20%. IAH and ACS are indicated primarily by intra-abdominal pressure (IAP) with IAH defined as IAP greater than 12 mmHg and ACS defined as IAP greater than 20 mmHg. This is often, but not exclusively, seen in neonates with necrotizing enterocolitis (NEC) (inflammation or perforation of the intestines). In neonates, even slightly elevated IAP of 5 mmHg, well before the threat of ACS, can lead to extreme discomfort and difficulty feeding as additional fluid introduced to the abdominal area from feeding further elevates IAP, dangerously progressing the neonate to ACS. Disrupting an infant’s nutritional intake is especially detrimental at this stage in life where regular feeding schedules must be maintained to help babies grow and develop. The gold standard of quantitative IAP measurements is a peritoneal catheter; however, catheterization is invasive and very risky and may require sedation or paralysis. As a result, physicians rely on palpation to monitor IAP. However, studies have shown that palpation is unreliable with up to 60% of patients being misdiagnosed. Given the need for monitoring IAP to prevent ACS, support regular nutrition, and detect NEC, the difficulty in monitoring IAP using a catheter, and the unreliability of palpation, there remains a need for a simple, noninvasive method of measuring IAP in neonatal populations.
Abdominal wall tension (AWT) has shown potential as a non-invasive, indirect measure of IAP, being previously validated in adults. AWT can be measured with a force sensor as the sensor lightly presses into the abdomen (~5 mm depth) and is similar in principle to the palpation method but provides objective measures of force instead of the “sensation of force” as with palpation. Previous studies have correlated AWT to IAP in adults, but not in neonates. Given the differences in skin and soft tissue elasticity of neonates compared to adults, which could impact the relationship between AWT and IAP, it is necessary to explicitly characterize the relationship between AWT and IAP in the neonates to assess whether AWT is a proper indirect measure of IAP in this population. This research aims to demonstrate that AWT is a safe, non-invasive, indirect measure of IAP in neonates and can be leveraged to decrease the incidence of IAH, ACS, and NEC, as well as support clinical decision-making regarding nutrition.





Post-operative monitoring has many difficulties since medical professionals can't actively track their patient's health. Patients who have undergone pancreatic surgery for cancer have a high readmission rate because of the complexity of the surgery. Pancreatic cancer surgery can entail the removal of parts of the small intestines, part of the bile duct, gall bladder, lymph nodes, spleen, parts of the pancreas, and even sometimes the whole pancreas. Patients who have gone through pancreatic cancer surgery are at higher risk of leaking from the reconnection of organs, infections, internal bleeding, and problems digesting food. These high-risk factors are why post-operative monitoring is essential in reducing the chances of readmission by catching issues early. Studies have shown the benefits of wearable devices by predicting the possibility of readmission by monitoring health data such as steps, heart rate, and sleep. Still, they have had difficulty in missing data due to improper adherence, running out of battery, or complications connecting to phones for data storage. Modern-day wearable devices can also be complicated for elderly patients because of their various applications or uses. There are also issues with clarifying FDA-approved settings within certain smartwatches. This research aims to develop a simple, easy to use wearable for frustration-free post-op monitoring of pancreatic cancer patients.
Replace this text component with your accordion's content.

We are all familiar with the sight of an athlete experiencing a severe blow to the head and being unable to stand or walking shakily back to the sideline. In those instances, it is clear that the athlete has experienced a “concussion.” Concussion, as defined by the Guideline Development Subcommittee of the American Academy of Neurology, is a syndrome of biomechanically induced alteration of brain function potentially affecting memory, orientation, and state of consciousness and is the most common type of mild traumatic brain injury. Despite early work suggesting that concussion symptoms were directly correlated with the magnitudes of singular impacts, a more complete analysis of the data has made it clear that an impact threshold does not exist. In fact, the deleterious consequences of repetitive head impacts or sub-concussive blows can accumulate over time, causing tissue level damage that both increases susceptibility to later head impacts, and generally increases the volume of damaged brain tissue.
While numerous attempts have been made to design sensor systems to measure head impacts, including in-helmet systems, wearable accelerometers, and even mouthguards, they only quantify risk of injury and even so with varying accuracy. Additionally, these sensors do little to encourage better technique. Recent work demonstrating that improved technique decreases an athlete’s exposure to head impacts suggests that the best intervention is one that helps coaches better prepare their athletes for practices and games. The goal of this project is to develop a system that mitigates head injury by tracking the force of each hit and provides a video-based tool that will allow coaches to teach better technique.
The student will work with a senior member of the Hoilett and Human Injury Research and Regenerative Technologies (HIRRT) labs to develop a miniaturized device that combines high-impact accelerometers, GPS, and Bluetooth capabilities into a single, discreet wearable. The wearable will track the force delivered by repetitive head impacts, map the location of these forces in real-time on a practice field, and relay this information back to a bay station that can be monitored by coaches and athletic training staff.
Protege Mentors
- All mentors have extensive research programs
- All mentors are known for working with young researchers
- Faculty usually have extensive research programs
- Have graduate students that help mentor Protege students
- Industry mentors have group members that help mentor students
Protege mentors are expected to work with beginning students to develop their research project. Mentors should provide students with background information about the project and be able to answer the following:
- What is the goal?
- What approach is envisioned?
- What results are being sought?
Protege mentors should provide resources required to accomplish the project and be available to provide ongoing advice on the project. They should personally spend about three (3) hours per week with the student.
Contact Us

Gautam Pillay
Associate Dean of Research, CEAS – Research Development
736 Rhodes Hall